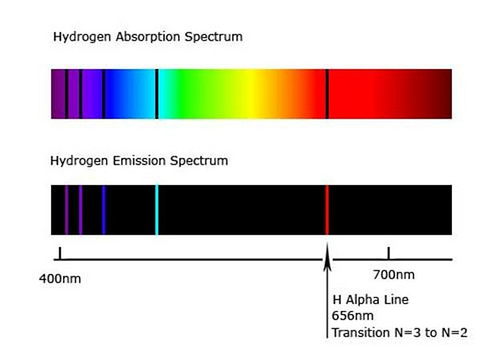
Colored lines appear against a black background in the emission. Because the energy levels are quantized, the spectrum for this transition is made up of lines. In the emission spectrum, when an excited electron goes to a lower energy level, it emits a certain amount of energy in the form of photons. Continuous spectra are produced by solids at high temperatures, such as the filament of electric light. Ques: What are the differences between the continuous spectrum and the emission spectrum?Īns: A continuous spectrum is one with no gaps in wavelengths over a wide range. When an incandescent object's light is resolved using a prism or spectroscope, it produces a continuous spectrum of colors.The ability of particular objects to hold heat and the quantity of absorption can be determined using absorption spectra.When energy is absorbed by electrons in the ground state to reach higher energy levels, an absorption spectrum is formed by the frequencies of light transmitted with dark bands.The frequencies of the released light, compose the emission spectrum. To return to ground states from the excited state, which is unstable, these excited electrons must emit energy. When an atom's electrons absorb energy, they migrate from lower energy levels to higher energy levels.Isaac Newton introduced the term spectrum to optics in the 17th century, referring to the range of colors seen when white light is scattered via a prism.The atomic emission spectrum can be used to identify elements because of these distinct properties. Similarly, indium causes a flame to turn blue when it is introduced into it. 
In the spectra of the Sun and other stars, absorption lines can be seen.įor example, the sodium atoms emit an amber yellow color when the platinum wire is immersed in a sodium nitrate solution and then placed into a flame. The composition of a given substance can be determined using emission spectra.įor example, in a star's surface layers or a planet's atmosphere. The ability of particular objects to hold heat and the quantity of absorption can be determined using absorption spectra. When atoms or molecules are de-excited from a higher to lower energy state, spectra are formed. When atoms or molecules are excited from a lower to a higher energy level, spectra are formed. On a black background, white lines or colored lines appear. 
On a white background, black lines appear. The emission spectrum is derived from the radiation emitted by the substances. When a substance absorbs radiation, it produces an absorption spectrum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
